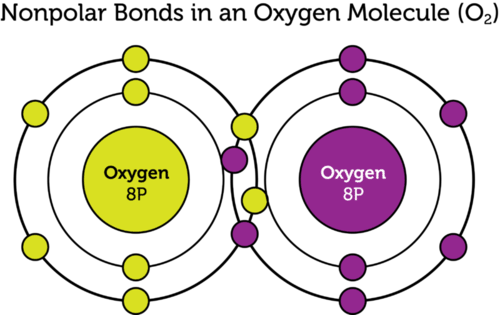
What Are Three Examples Of Nonpolar Covalent Bonds
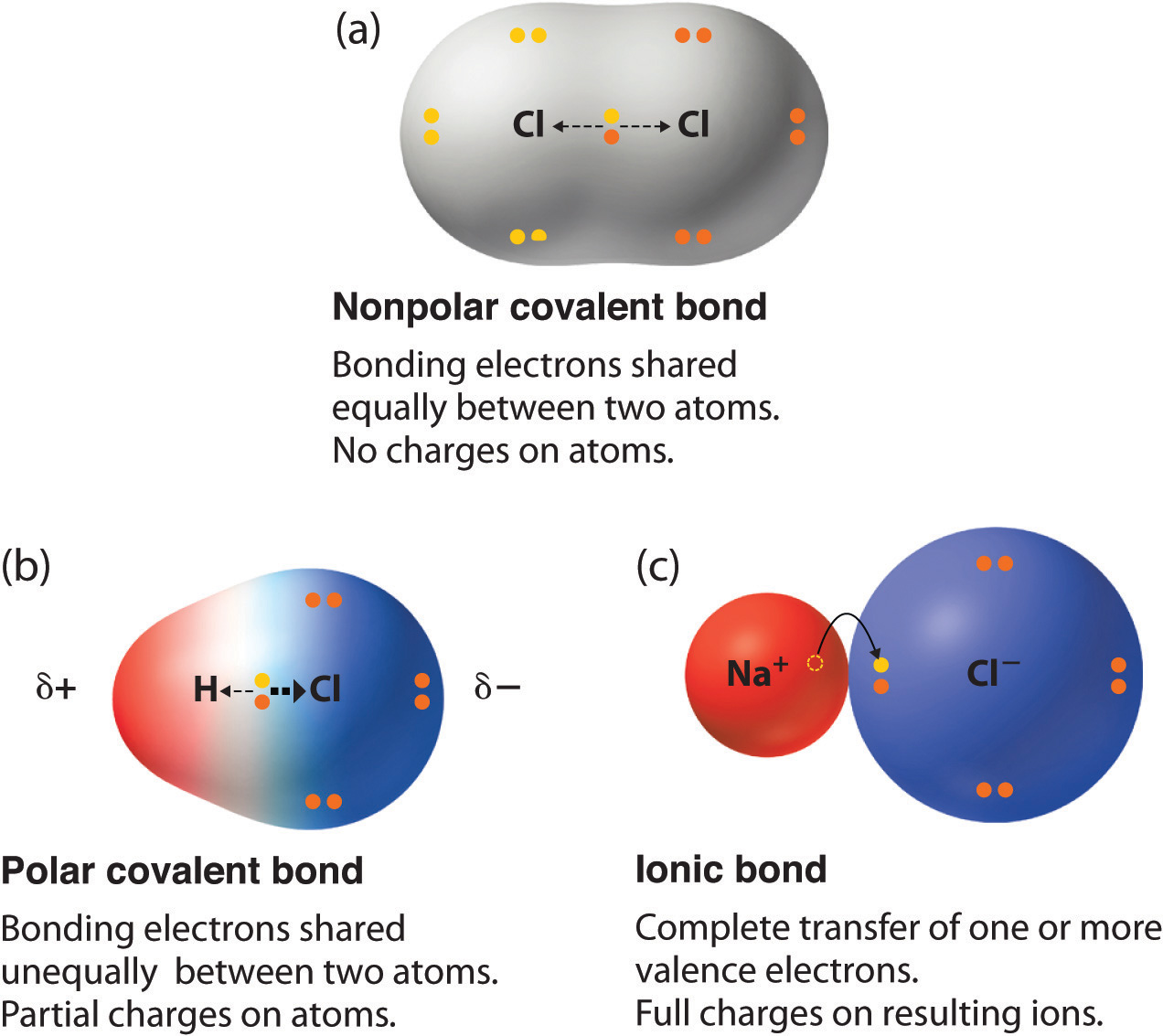
This partial ionic character of covalent bonds increases with the difference in the electronegativities of the two. Nonpolar Covalent Bonds.

Covalent Bond Definition Properties Examples Facts Britannica
Thus as the electronegativity difference of the two bonded elements increases a nonpolar bond gives way to a polar bond which in turn becomes an ionic bond.

What are three examples of nonpolar covalent bonds. It is important to know how many bonds certain elements will form in order to draw structural. Non-Covalent Bonds The backbone and side chain bonds are all covalent bonds as are disulfide bonds but non-covalent bonds are required to maintain secondary tertiary and quaternary structure These include Hydrogen bonds H-bonds Electrostatic Salt Bridges van der Waals. A nonpolar covalent bond is a covalent bond in which the bonding electrons are shared equally between the two.
When a polyatomic ion forms an ionic bond with another ion a polyatomic ionic compound is made. There are in fact no purely ionic bonds just as there are no purely covalent bonds. A bond in which the electronegativity difference is less than 19 is considered to be mostly covalent in character.
The number of covalent bonds an atom can form relates to the number of electrons it can share and still result in a neutral molecule. For example the 1 barium ion can form an ionic bond with the -1 hydroxide ion to form the Barium Hydroxide. However at this point we need to distinguish between two general types of covalent bonds.
Covalent bonds between identical atoms as in H 2 are nonpolarie electrically uniformwhile those between unlike atoms are polarie one atom is slightly negatively charged and the other is slightly positively charged. Polyatomic ions are formed when a group of atoms have a charge. Bonding is a continuum of types.
Hydroxide for example is formed when oxygen and hydrogen covalently bond but still have a charge of -1. Carbon forms 4 covalent bonds in neutral compounds Covalent bonding involves the sharing of electrons between atoms.
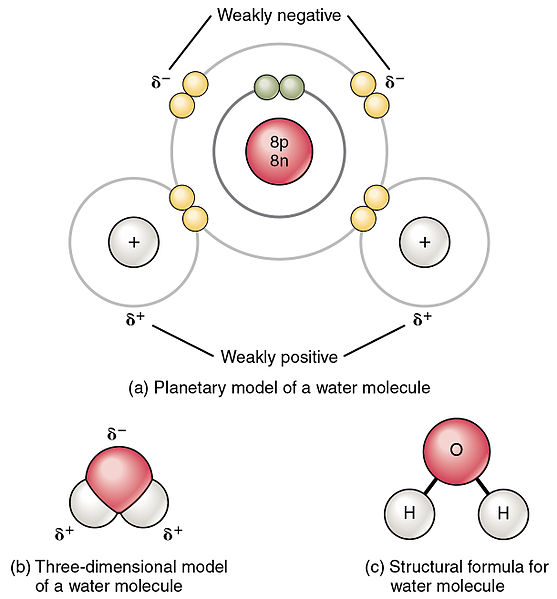
Polar And Non Polar Molecules Vce Chemistry

The Bonds S S O H And Ca Cl Are Respectively A Nonpolar Covalent Polar Covalent Ionic B Polar Covalent Nonpolar Covalent Ionic C Ionic Polar Covalent Nonpolar Covalent D Nonpolar Covalent Study Com

Bond Polarity Chemistry For Non Majors
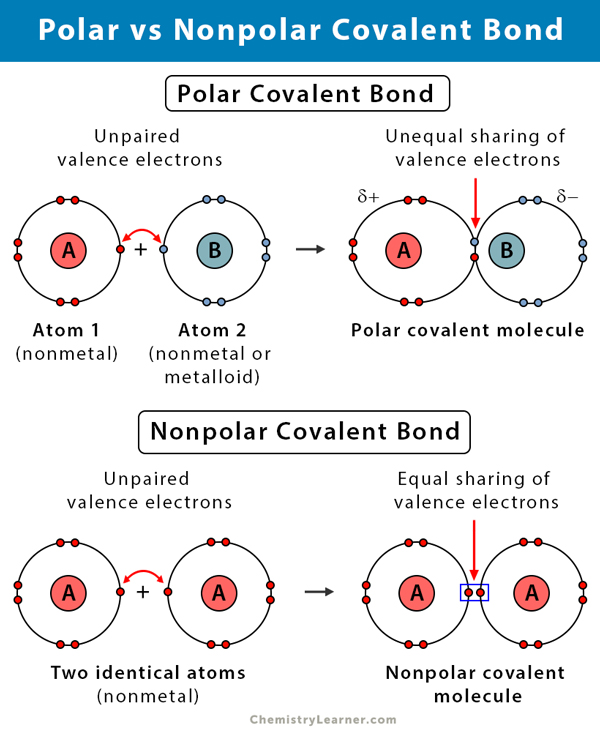
Polar Covalent Bond Definition And Examples
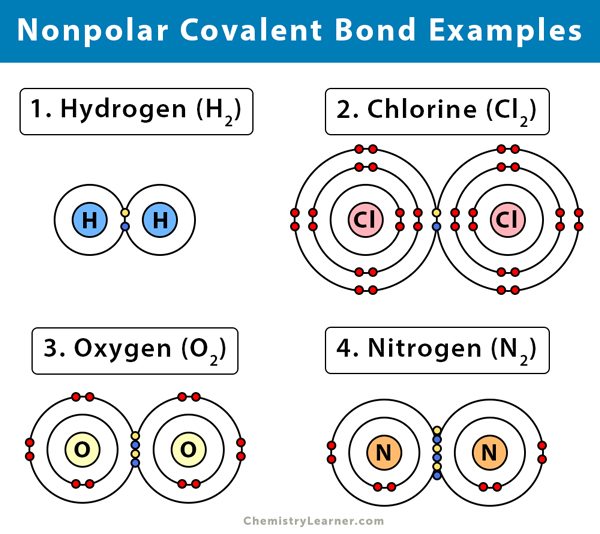
Nonpolar Covalent Bond Definition And Examples
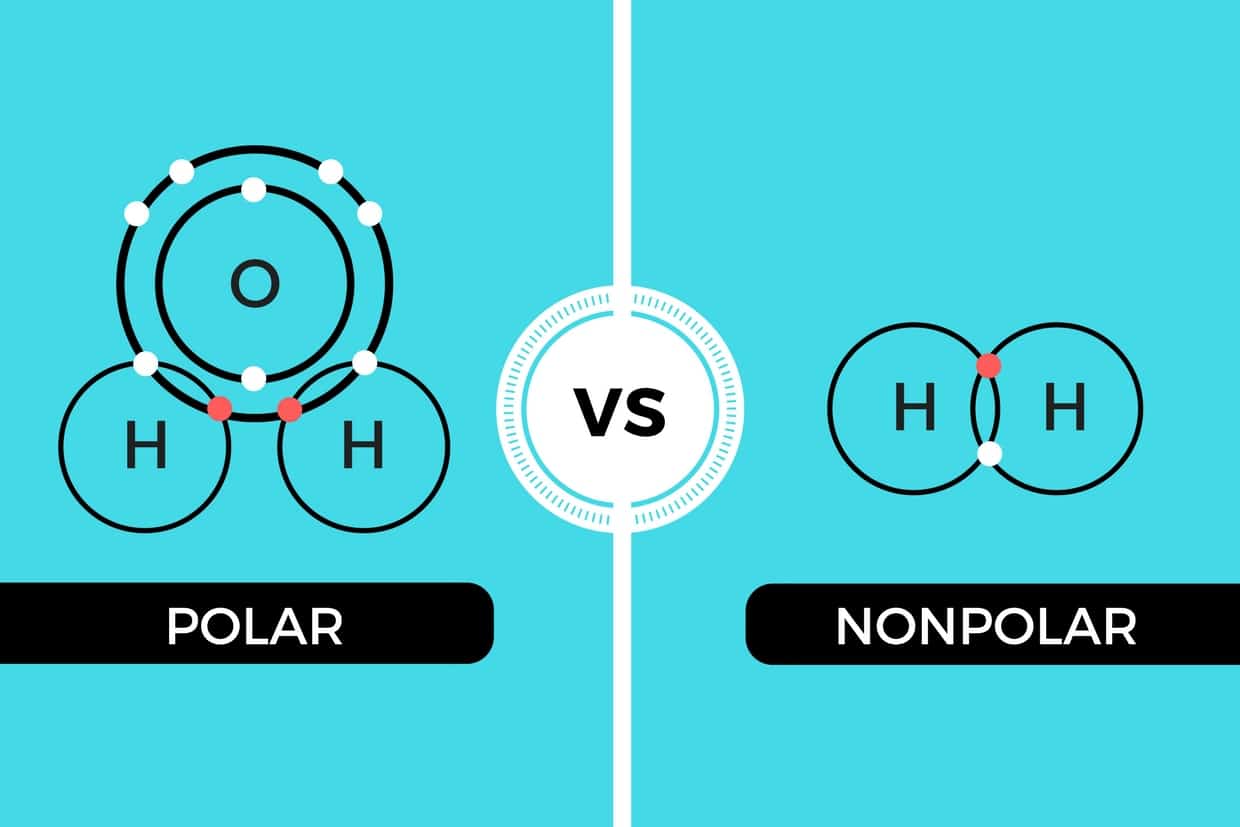
Polar Vs Nonpolar It S All About Sharing On An Atomic Level
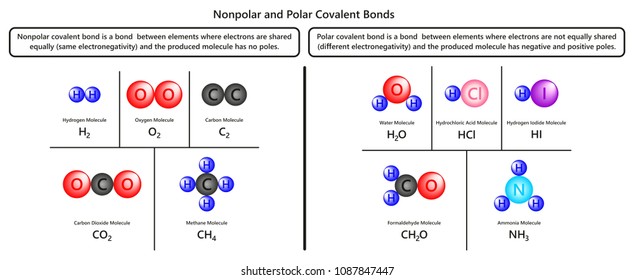
Nonpolar Images Stock Photos Vectors Shutterstock
Difference Between Non Polar And Polar Covalent Bonds Difference Between
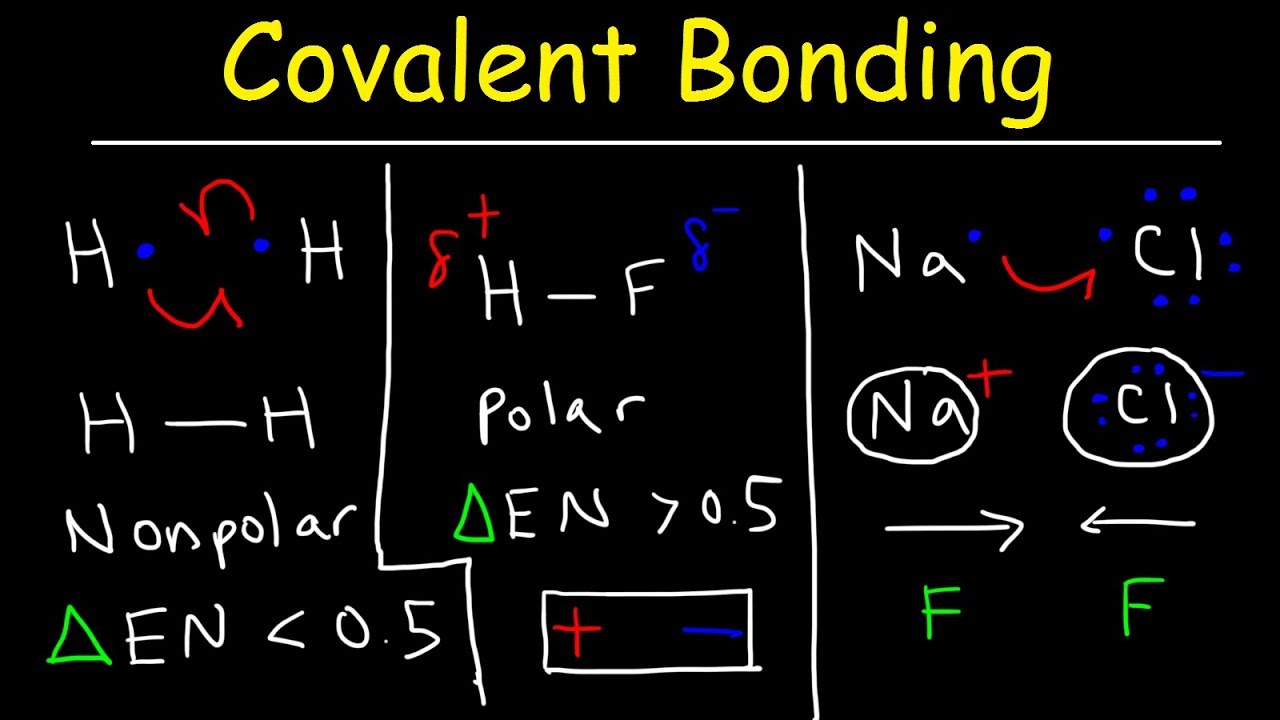
Polar Covalent Bonds And Nonpolar Covalent Bonds Ionic Bonding Types Of Chemical Bonds Youtube

Chemical Bonds Mhcc Biology 112 Biology For Health Professions
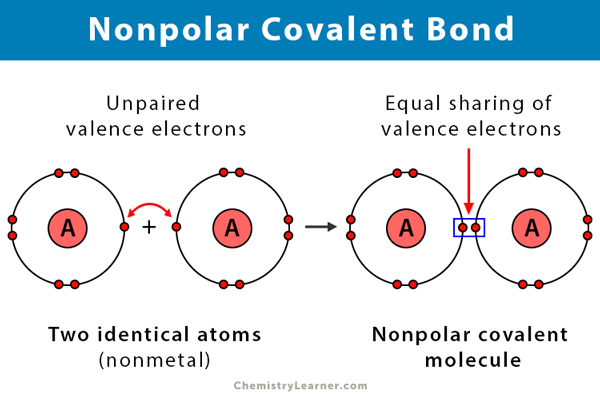
Nonpolar Covalent Bond Definition And Examples
Covalent Bonds Open Textbooks For Hong Kong
Covalent Bond Definition Types Polar And Non Polar Covalent Bond Explained

Covalent Bonds Biology For Majors I
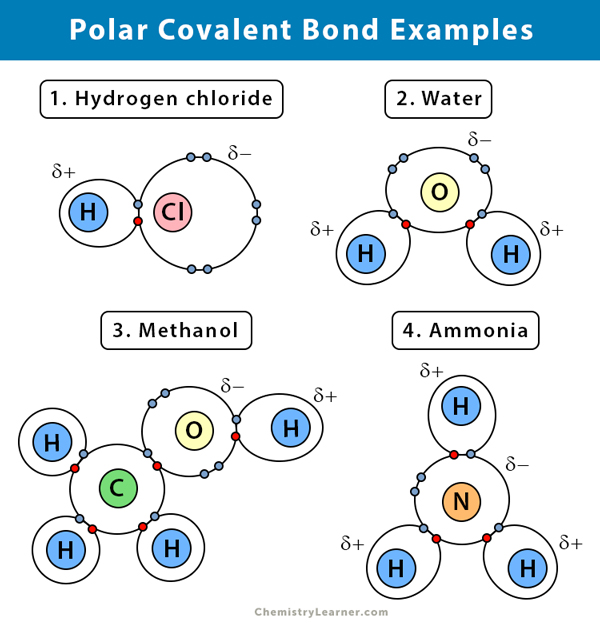
Polar Covalent Bond Definition And Examples
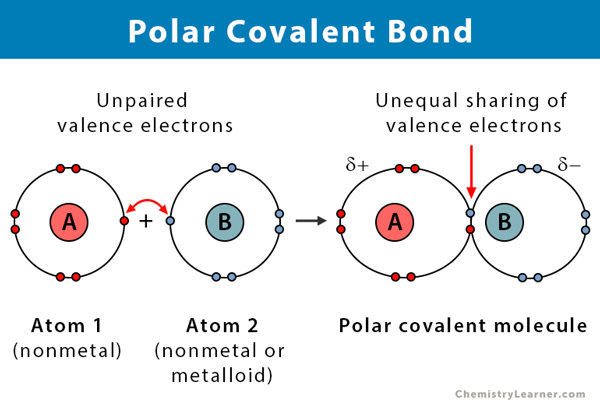
Polar Covalent Bond Definition And Examples

Polar And Nonpolar Covalent Bonds Definitions Molecules And Examples